HPLC columns contain a stationary phase (usually porous silica particles) bonded to a support material to provide a large surface area. The stationary phase provides the basis for the separation of sample components.
The stationary phase chemistry determines the affinity with which sample components adhere or are retained to the column as the sample flows through the column with the mobile phase. As a result, sample components move through the column and elute at different rates.
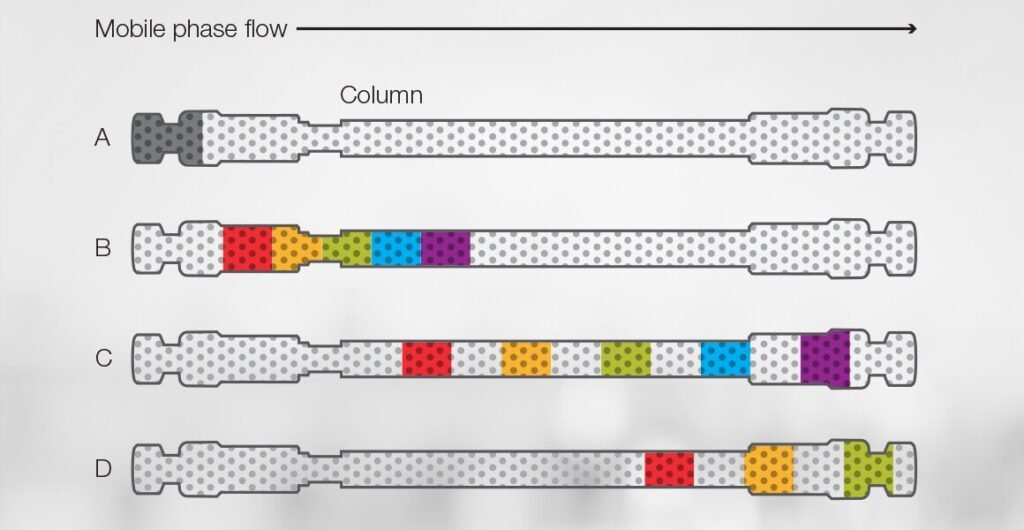
For example, you can visualize a mixture containing multiple components (A) as gray bands appearing at the front or head of the column.
As the mixture flows through the column with the mobile phase, the red component (B) is more retained than the purple component. Therefore, the purple and blue components move through the column faster and are the first two “bands” to elute from the column (C). The green, yellow, and red bands are retained longer and elute later (D).